Courses tagged with "Diencephalon" (35)
CHEM 216 builds on the experimental approach started in CHEM 211. Students participate in planning exactly what they are going to do in the laboratory by being given general goals and directions that have to be adapted to fit the specific project they will be working on. They use microscale equipment, which requires them to develop manual dexterity and care in working in the laboratory. They also evaluate the results of their experiments by checking for identity and purity using various chromatographic and spectroscopic methods. Course Level: Undergraduate This Work, Chemistry 216 - The Synthesis and Characterization of Carbonyl Compounds, by Ginger Shultz is licensed under a Creative Commons Attribution-ShareAlike license.
Organic Chemistry of Macromolecules covers the preparation, reactions, and properties of high molecular weight polymeric materials of both natural and synthetic origin. As a part of this course, U-M students collaboratively created and edited Wikipedia articles. Course Level: Graduate This Work, Chemistry 538 - Organic Chemistry of Macromolecules, by Anne McNeil is licensed under a Creative Commons Attribution-ShareAlike license.
Acid Base Introduction. pH, pOH of Strong Acids and Bases. pH of a Weak Acid. pH of a Weak Base. Conjugate Acids and Bases. pKa and pKb Relationship. Buffers and Hendersen-Hasselbalch. Strong Acid Titration. Weak Acid Titration. Half Equivalence Point. Titration Roundup. Acid Base Titration. Acid Base Introduction. pH, pOH of Strong Acids and Bases. pH of a Weak Acid. pH of a Weak Base. Conjugate Acids and Bases. pKa and pKb Relationship. Buffers and Hendersen-Hasselbalch. Strong Acid Titration. Weak Acid Titration. Half Equivalence Point. Titration Roundup. Acid Base Titration.
Molecular and Empirical Formulas. The Mole and Avogadro's Number. Formula from Mass Composition. Another mass composition problem. Balancing Chemical Equations. Stoichiometry. Stoichiometry Example Problem 1. Stoichiometry Example Problem 2. Stoichiometry: Limiting Reagent. Limiting Reactant Example Problem 1. Spectrophotometry Introduction. Spectrophotometry Example. Molecular and Empirical Formulas. The Mole and Avogadro's Number. Formula from Mass Composition. Another mass composition problem. Balancing Chemical Equations. Stoichiometry. Stoichiometry Example Problem 1. Stoichiometry Example Problem 2. Stoichiometry: Limiting Reagent. Limiting Reactant Example Problem 1. Spectrophotometry Introduction. Spectrophotometry Example.
Ideal Gas Equation: PV=nRT. Ideal Gas Equation Example 1. Ideal Gas Equation Example 2. Ideal Gas Equation Example 3. Ideal Gas Equation Example 4. Partial Pressure. Vapor Pressure Example. Ideal Gas Equation: PV=nRT. Ideal Gas Equation Example 1. Ideal Gas Equation Example 2. Ideal Gas Equation Example 3. Ideal Gas Equation Example 4. Partial Pressure. Vapor Pressure Example.
Elements and Atoms. Introduction to the atom. Elements and Atoms. Introduction to the atom.
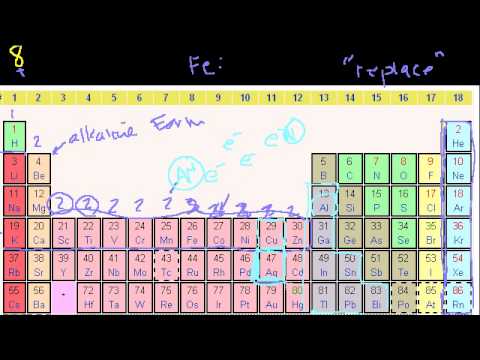
Orbitals. More on orbitals and electron configuration. Electron configurations. Electron configurations 2. Valence Electrons. Orbitals. More on orbitals and electron configuration. Electron configurations. Electron configurations 2. Valence Electrons.
Introduction to Oxidation States. More on Oxidation States. Hydrogen Peroxide Correction. Redox Reactions. Galvanic Cells. Introduction to Oxidation States. More on Oxidation States. Hydrogen Peroxide Correction. Redox Reactions. Galvanic Cells.
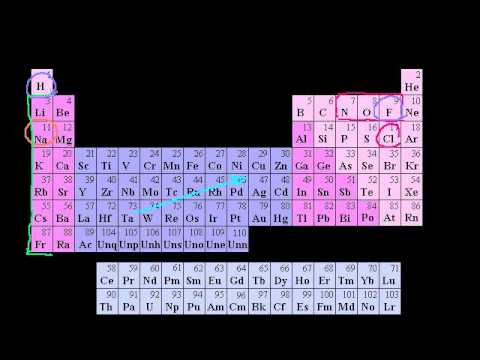
Groups of the Periodic Table. Valence Electrons. Periodic Table Trends: Ionization Energy. Other Periodic Table Trends. Ionic, Covalent, and Metallic Bonds. Groups of the Periodic Table. Valence Electrons. Periodic Table Trends: Ionization Energy. Other Periodic Table Trends. Ionic, Covalent, and Metallic Bonds.
Types of Decay. Half-Life. Exponential Decay Formula Proof (can skip, involves Calculus). Introduction to Exponential Decay. More Exponential Decay Examples. Types of Decay. Half-Life. Exponential Decay Formula Proof (can skip, involves Calculus). Introduction to Exponential Decay. More Exponential Decay Examples.
Introduction to Kinetics. Reactions in Equilibrium. Mini-Video on Ion Size. Keq Intuition (mathy and not necessary to progress). Keq derivation intuition (can skip; bit mathy). Heterogeneous Equilibrium. Le Chatelier's Principle. Introduction to pH, pOH, and pKw. Introduction to Kinetics. Reactions in Equilibrium. Mini-Video on Ion Size. Keq Intuition (mathy and not necessary to progress). Keq derivation intuition (can skip; bit mathy). Heterogeneous Equilibrium. Le Chatelier's Principle. Introduction to pH, pOH, and pKw.
States of Matter. States of Matter Follow-Up. Specific Heat, Heat of Fusion and Vaporization. Chilling Water Problem. Phase Diagrams. Van Der Waals Forces. Covalent Networks, Metallic, and Ionic Crystals. Vapor Pressure. Suspensions, Colloids and Solutions. Solubility. Boiling Point Elevation and Freezing Point Suppression. Change of State Example. States of Matter. States of Matter Follow-Up. Specific Heat, Heat of Fusion and Vaporization. Chilling Water Problem. Phase Diagrams. Van Der Waals Forces. Covalent Networks, Metallic, and Ionic Crystals. Vapor Pressure. Suspensions, Colloids and Solutions. Solubility. Boiling Point Elevation and Freezing Point Suppression. Change of State Example.
Chemistry is an integral part of our lives and the world we live in. Chemistry explains the world around us. Are you a college student intimidated by a chemistry course? Do you need a head start in exploring chemistry in order to be prepared for general chemistry courses? In this pre-college course, students will be introduced to the fundamentals of chemistry. Concepts, terminologies, and basic mathematics skills required for conversions in chemistry will be covered. This basic chemistry course is recommended for McHenry County College
This course provides an introduction to the chemistry of biological, inorganic, and organic molecules. The emphasis is o
Моделирование биологичеÑких молекул - одна из бурно развивающихÑÑ Ð¾Ð±Ð»Ð°Ñтей Ñовременной науки. Ð’ курÑе даютÑÑ Ð¾Ñновы ÑÑ‚Ñ€Ð¾ÐµÐ½Ð¸Ñ Ð±Ð¸Ð¾Ð¼Ð¾Ð»ÐµÐºÑƒÐ», примеры Ð¿Ñ€Ð¸Ð¼ÐµÐ½ÐµÐ½Ð¸Ñ Ð¿Ñ€Ð¾Ð³Ñ€Ð°Ð¼Ð¼Ð½Ñ‹Ñ… пакетов Ð´Ð»Ñ Ð¼Ð¾Ð»ÐµÐºÑƒÐ»Ñрного моделированиÑ, разъÑÑнÑÑŽÑ‚ÑÑ Ð¿Ð¾Ð´Ñ…Ð¾Ð´Ñ‹ к математичеÑкому опиÑанию молекулÑрных ÑиÑтем и разбираетÑÑ Ð¿Ñ€Ð¾Ð³Ñ€Ð°Ð¼Ð¼Ð½Ð°Ñ Ñ€ÐµÐ°Ð»Ð¸Ð·Ð°Ñ†Ð¸Ñ Ñтих подходов на центральном и графичеÑком процеÑÑорах.
Trusted paper writing service WriteMyPaper.Today will write the papers of any difficulty.