Courses tagged with "Diencephalon" (58)
If chemistry is the science of stuff, then analytical chemistry answers the question: what is it? And how much of it do you have? This course teaches how to do this with instrumental analysis!
This introduction to fundamental chemical concepts of atomic and molecular structure will emphasize the development of these concepts from experimental observations and scientific reasoning.
Elements and Atoms. Introduction to the atom. Elements and Atoms. Introduction to the atom.
Orbitals. More on orbitals and electron configuration. Electron configurations. Electron configurations 2. Valence Electrons. Orbitals. More on orbitals and electron configuration. Electron configurations. Electron configurations 2. Valence Electrons.
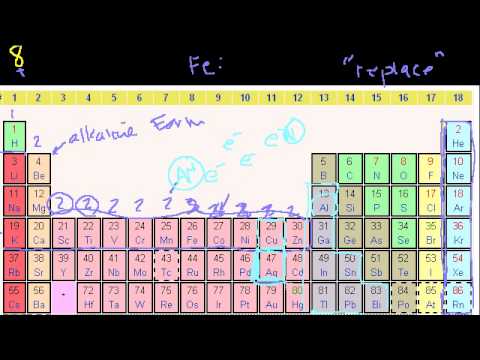
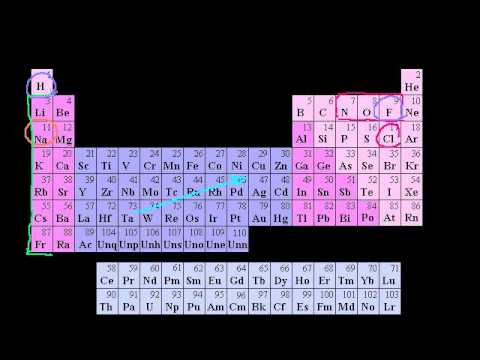
Groups of the Periodic Table. Valence Electrons. Periodic Table Trends: Ionization Energy. Other Periodic Table Trends. Ionic, Covalent, and Metallic Bonds. Groups of the Periodic Table. Valence Electrons. Periodic Table Trends: Ionization Energy. Other Periodic Table Trends. Ionic, Covalent, and Metallic Bonds.
Molecular and Empirical Formulas. The Mole and Avogadro's Number. Formula from Mass Composition. Another mass composition problem. Balancing Chemical Equations. Stoichiometry. Stoichiometry Example Problem 1. Stoichiometry Example Problem 2. Stoichiometry: Limiting Reagent. Limiting Reactant Example Problem 1. Spectrophotometry Introduction. Spectrophotometry Example. Molecular and Empirical Formulas. The Mole and Avogadro's Number. Formula from Mass Composition. Another mass composition problem. Balancing Chemical Equations. Stoichiometry. Stoichiometry Example Problem 1. Stoichiometry Example Problem 2. Stoichiometry: Limiting Reagent. Limiting Reactant Example Problem 1. Spectrophotometry Introduction. Spectrophotometry Example.
Ideal Gas Equation: PV=nRT. Ideal Gas Equation Example 1. Ideal Gas Equation Example 2. Ideal Gas Equation Example 3. Ideal Gas Equation Example 4. Partial Pressure. Vapor Pressure Example. Ideal Gas Equation: PV=nRT. Ideal Gas Equation Example 1. Ideal Gas Equation Example 2. Ideal Gas Equation Example 3. Ideal Gas Equation Example 4. Partial Pressure. Vapor Pressure Example.
States of Matter. States of Matter Follow-Up. Specific Heat, Heat of Fusion and Vaporization. Chilling Water Problem. Phase Diagrams. Van Der Waals Forces. Covalent Networks, Metallic, and Ionic Crystals. Vapor Pressure. Suspensions, Colloids and Solutions. Solubility. Boiling Point Elevation and Freezing Point Suppression. Change of State Example. States of Matter. States of Matter Follow-Up. Specific Heat, Heat of Fusion and Vaporization. Chilling Water Problem. Phase Diagrams. Van Der Waals Forces. Covalent Networks, Metallic, and Ionic Crystals. Vapor Pressure. Suspensions, Colloids and Solutions. Solubility. Boiling Point Elevation and Freezing Point Suppression. Change of State Example.
Introduction to Oxidation States. More on Oxidation States. Hydrogen Peroxide Correction. Redox Reactions. Galvanic Cells. Introduction to Oxidation States. More on Oxidation States. Hydrogen Peroxide Correction. Redox Reactions. Galvanic Cells.
Types of Decay. Half-Life. Exponential Decay Formula Proof (can skip, involves Calculus). Introduction to Exponential Decay. More Exponential Decay Examples. Types of Decay. Half-Life. Exponential Decay Formula Proof (can skip, involves Calculus). Introduction to Exponential Decay. More Exponential Decay Examples.
This course provides an introduction to the chemistry of biological, inorganic, and organic molecules. The emphasis is o
Introduction to Kinetics. Reactions in Equilibrium. Mini-Video on Ion Size. Keq Intuition (mathy and not necessary to progress). Keq derivation intuition (can skip; bit mathy). Heterogeneous Equilibrium. Le Chatelier's Principle. Introduction to pH, pOH, and pKw. Introduction to Kinetics. Reactions in Equilibrium. Mini-Video on Ion Size. Keq Intuition (mathy and not necessary to progress). Keq derivation intuition (can skip; bit mathy). Heterogeneous Equilibrium. Le Chatelier's Principle. Introduction to pH, pOH, and pKw.
Acid Base Introduction. pH, pOH of Strong Acids and Bases. pH of a Weak Acid. pH of a Weak Base. Conjugate Acids and Bases. pKa and pKb Relationship. Buffers and Hendersen-Hasselbalch. Strong Acid Titration. Weak Acid Titration. Half Equivalence Point. Titration Roundup. Acid Base Titration. Acid Base Introduction. pH, pOH of Strong Acids and Bases. pH of a Weak Acid. pH of a Weak Base. Conjugate Acids and Bases. pKa and pKb Relationship. Buffers and Hendersen-Hasselbalch. Strong Acid Titration. Weak Acid Titration. Half Equivalence Point. Titration Roundup. Acid Base Titration.
This course deals with a more advanced treatment of the biochemical mechanisms that underlie biological processes. Emphasis will be given to the experimental methods used to unravel how these processes fit into the cellular context as well as the coordinated regulation of these processes. Topics include macromolecular machines for energy and force transduction, regulation of biosynthetic and degradative pathways, and the structure and function of nucleic acids.
5.451 is a half-semester introduction to natural product biosynthetic pathways. The course covers the assembly of complex polyketide, peptide, terpene and alkaloid structures. Discussion topics include chemical and biochemical strategies used to elucidate natural product pathways.
This is the first semester in a two-semester introductory course focused on current theories of structure and mechanism in organic chemistry, their historical development, and their basis in experimental observation. The course is open to freshmen with excellent preparation in chemistry and physics, and it aims to develop both taste for original science and intellectual skills necessary for creative research.
This is a continuation of Freshman Organic Chemistry I (CHEM 125a), the introductory course on current theories of structure and mechanism in organic chemistry for students with excellent preparation in chemistry and physics. This semester treats simple and complex reaction mechanisms, spectroscopy, organic synthesis, and some molecules of nature.
This course is an intensive introduction to the techniques of experimental chemistry and gives first year students an opportunity to learn and master the basic chemistry lab techniques for carrying out experiments. Students who successfully complete the course and obtain a "Competent Chemist" (CC) or "Expert Experimentalist" (EE) rating are likely to secure opportunities for research work in a chemistry lab at MIT.
Acknowledgements
The laboratory manual and materials for this course were prepared by Dr. Katherine J. Franz and Dr. Kevin M. Shea with the assistance of Professors Rick L. Danheiser and Timothy M. Swager. Materials have been revised by Dr. J. Haseltine, Dr. Kevin M. Shea, Dr. Sarah A. Tabacco, Dr. Kimberly L. Berkowski, Anne M. (Gorham) Rachupka, and Dr. John J. Dolhun.
WARNING NOTICE
The experiments described in these materials are potentially hazardous and require a high level of safety training, special facilities and equipment, and supervision by appropriate individuals. You bear the sole responsibility, liability, and risk for the implementation of such safety procedures and measures. MIT shall have no responsibility, liability, or risk for the content or implementation of any of the material presented.
Legal Notice
This course deals with the application of structure and theory to the study of organic reaction mechanisms: Stereochemical features including conformation and stereoelectronic effects; reaction dynamics, isotope effects and molecular orbital theory applied to pericyclic and photochemical reactions; and special reactive intermediates including carbenes, carbanions, and free radicals.
Organic Chemistry of Macromolecules covers the preparation, reactions, and properties of high molecular weight polymeric materials of both natural and synthetic origin. As a part of this course, U-M students collaboratively created and edited Wikipedia articles. Course Level: Graduate This Work, Chemistry 538 - Organic Chemistry of Macromolecules, by Anne McNeil is licensed under a Creative Commons Attribution-ShareAlike license.
Trusted paper writing service WriteMyPaper.Today will write the papers of any difficulty.